|
Calculate the number of protons, neutrons and electrons it contains. The number of protons is the atomic number, and the. The atomic number of a sodium atom is 11 and its mass number is 23. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. The positively charged protons tend to repel each other, and the neutrons help to hold the nucleus together. This is the standard unit for atomic or molecular mass, and 1 amu is thus 1/12 th the mass of a 12 C atom. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The periodic tables arrangement also allows scientists to discern trends in element properties, including electronegativity, ionization energy, and atomic. The atomic mass unit (u or amu) is a relative unit based on a carbon-12 atom with six protons and six neutrons, which is assigned an exact value of 12 amu's (u's). The first chemical element is Hydrogen and the last is Ununoctium. The first example occurs in the case of the lanthanoids (elements having atomic numbers between 57 and 71).The lanthanoids have the general electron configuration Kr4d 10 4f i 5s 2 5p 6 5d 0 or 1 6s 2. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. Periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. The third major category of elements arises when the distinguishing electron occupies an f subshell. The periodic table - Edexcel Ionic compounds. The atomic number of an element is the number of. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. The numbers at the side of an element in the periodic table represent its atomic number and atomic mass. This list contains the 118 elements of chemistry. the atomic number of each element is unique. Separation and Concentration Purification RequestĬhemical elements listed by atomic number The elements of the periodic table sorted by atomic numberĬlick on any elements name for further chemical properties, environmental data or health effects.Plant Inspection & Process Optimalisation.The classification of elements by atomic number allows us to understand many properties of the atom and makes it possible to predict behaviors instead of just having to memorize everything. For example, carbon-12, carbon-13, and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13, and 14, respectively. 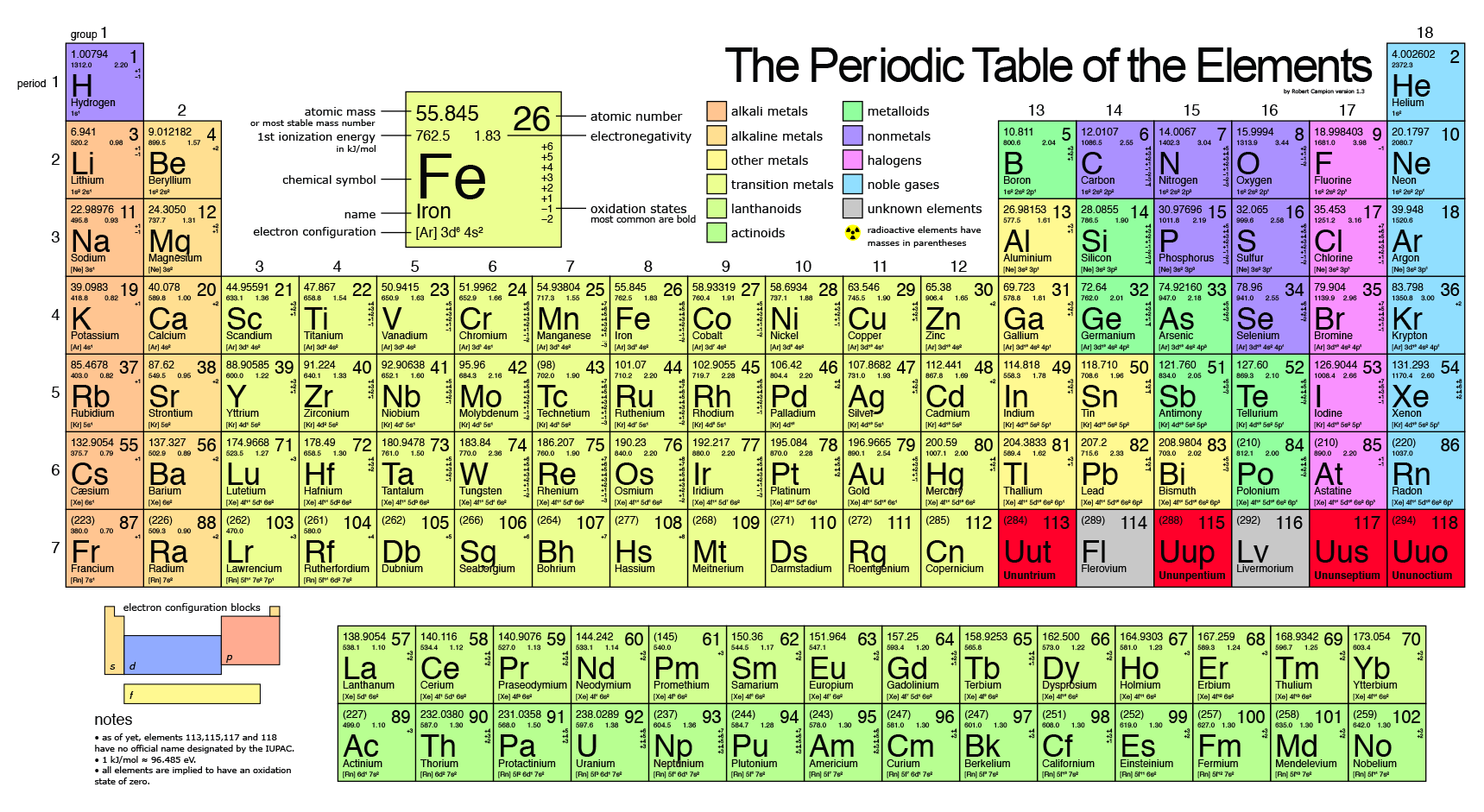
(Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain) The number of nucleons (both protons and neutrons) in the nucleus is the atoms mass number, and each isotope of a given element has a different mass number. \): The periodic table classifies elements by atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
